Research Philosophy
The explosive growth of NMR spectroscopy during the
last five decades is reflected in the vast number of
applications. Nowadays, NMR spectroscopy represents the
most versatile and informative technique for the
elucidation of structures, dynamics and kinetics in
solution. However, this rapid development means that
only a fraction of the analytical potential offered by
modern NMR spectroscopy is usually exploited by the
research chemists. Thus, I try to participate in
various scientific projects as a NMR specialist
encouraging and research chemists and students to
utilise modern NMR techniques. In former times, my work
involved scientific projects dealing mainly with
organotin compounds. Some of them are described below.
Now, I cooperate with various researchers within the
School of Chemistry, the Centre of Bimolecular Sciences
and Sasol Technology Research Laboratory. Therefore my
field of interest is widening and involves the organic
chemistry, biochemistry and homogeneous catalysis.
(3-Methoxypropyl)stannanes
Organotin compounds have revealed a wide spectrum of
biological merits. Antitumor properties in vitro
against a wide panel of tumour cell lines of human
origin are rather well known. It has been proposed that
anticancer reagents could also be used against
trypanosomal diseases (human sleeping sickness and
domesticated live stock diseases). Recently, it was
found that several organotin thiolates exhibit high in
vitro levels of activity compared with the arsenic
derivatives used for chemotherapy. However, antitumor
and trypanocidal efficiency of organotin derivatives
seems to be limited by their low water solubility.
Various sets of (3-methoxypropyl)stannanes have been
prepared as organotin compounds with considerable water
solubility. Although no or only negligible antitumor
activity was found, promising trypanocidal activity was
observed for some (3-methoxypropyl)stannanes.
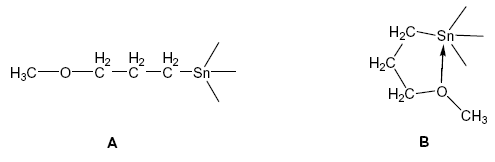
The 3-methoxypropyl group can be bonded either as
monodentate ligand (A) or as a bidentate
C,O-chelating ligand (B).
To distinguish between those two binding modes in
solution might be a crucial issue of structural
research. In order to find a indicator of
existence and eventually strength of O-Sn coordination,
structures of (3-methoxypropylstannanes) were studied
using X-ray crystallography,
119Sn CP/MAS NMR in the solid
state and by 1H,
13C,
17O,
119Sn NMR in solutions of
non-coordinating solvent (CDCl
3) and coordinating solvent
(dmso-d6). J(
1H,
119Sn) Coupling constants,
crucial to the study, were acquired by 1D
1H,
119Sn HMQC and 2D
1H,
119Sn J-HMBC heteronuclear
correlations.
2-Functionalised Vinylstannanes
Organometallics containing not only M-C bonds as
reactive sites but also other centres of high
reactivity represent a very interesting field of
organometallic chemistry. Introduction of a heteroatom
Y (Y is a Group 14-17 element) into an organoligand can
have a profound influence on the structure, stability
and reactivity of organometallic compounds and
consequently open the possibility of entirely new
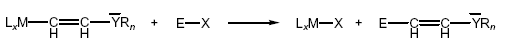
reactions. If organometallic compounds with
Lewis-basic 2-functionalised ethyl ligands
LxM─CH
2─CH
2YR
n (YR
n = NR
2, OR, Cl, …; R = alkyl, aryl,
H) are treated with suitable electrophiles, they can
undergo elimination reaction, so-called heterolytic
fragmentation.
On the other hand, in organotin compounds with
2-substituted vinyl ligands R'3SnCH=CHYRn, the electrophile attacks
preferably at the α-carbon atom and a cleavage of the
Sn-C bond takes place where significant difference in
reactivity of stereoisomers is observed.
Kinetic studies were carried out using
1H NMR.
1H-
119Sn HMQC spectra were employed
to analyse complex mixtures of products.